Pharma
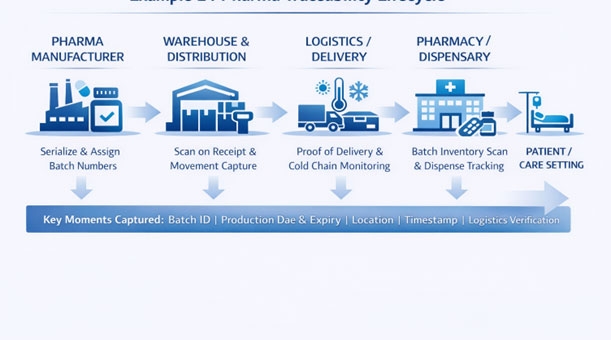
From manufacturing to pharmacy dispensary, Level 4 (L4) traceability provides batch-level track & trace across the end-to-end pharmaceutical supply chain — enhancing safety, compliance, and operational control. It enables organizations to capture, manage, and act on traceability data from production lines through distribution and on to the point of care.

What L4 Traceability Means in Pharma
This aligns with industry traceability expectations where companies must comply with traceability regulations , secure supply channels, manage recalls efficiently, and authenticate products throughout the chain.
L4 traceability is defined by:
L4 traceability is defined by:
Batch/lot tracking of medicines and medical products
Standardized identification & data capture at key checkpoints
Visibility across partners — manufacturers, distributors, pharmacies, and hospitals
Rapid recall and compliance capability in regulated environments